Interferon-producing universal sarbecovirus vaccines, and uses thereof
US20240139307
2024-05-02
Human necessities
A61K39/215
Inventor:
Assignee:
Applicant:
Drawings (4 of 28)
Smart overview of the Invention
The invention focuses on universal sarbecovirus vaccines that express interferon, specifically designed to combat various sarbecoviruses such as SARS-CoV-1, SARS-CoV-2, and its variants. By encoding interferon directly into the viral genome, the vaccine enhances mucosal T cell responses, thereby providing robust mucosal and heterotypic immunity. This approach aims to address the limitations of existing vaccines that struggle with new variants like omicron due to significant mutations in the spike protein.
Technological Innovation
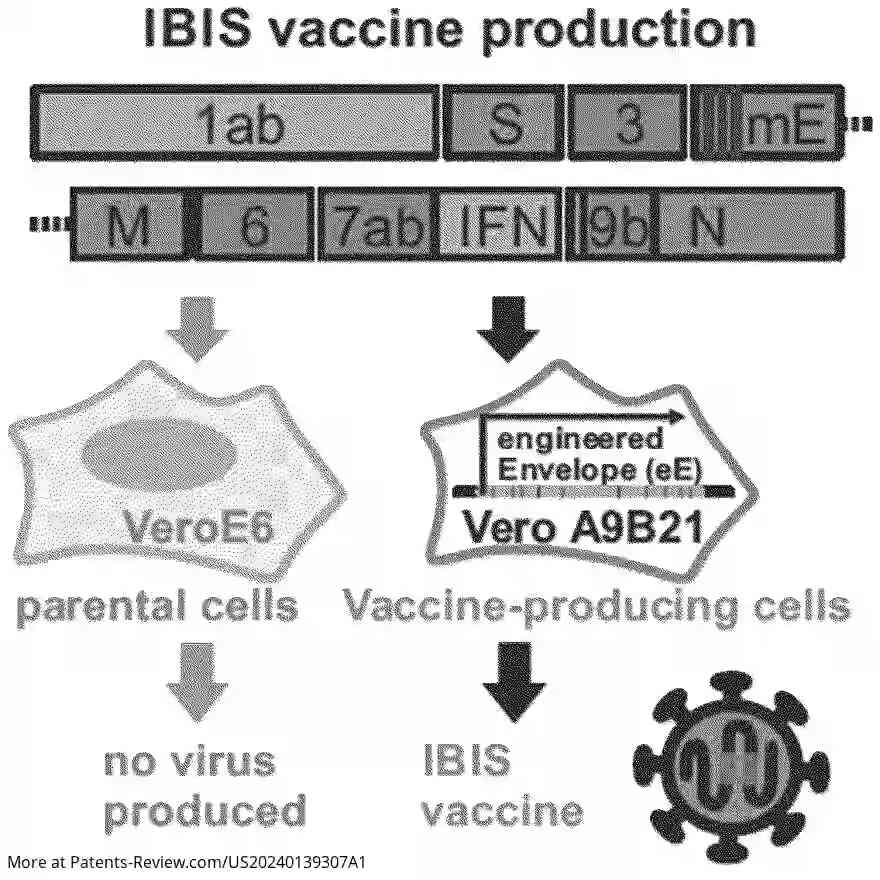
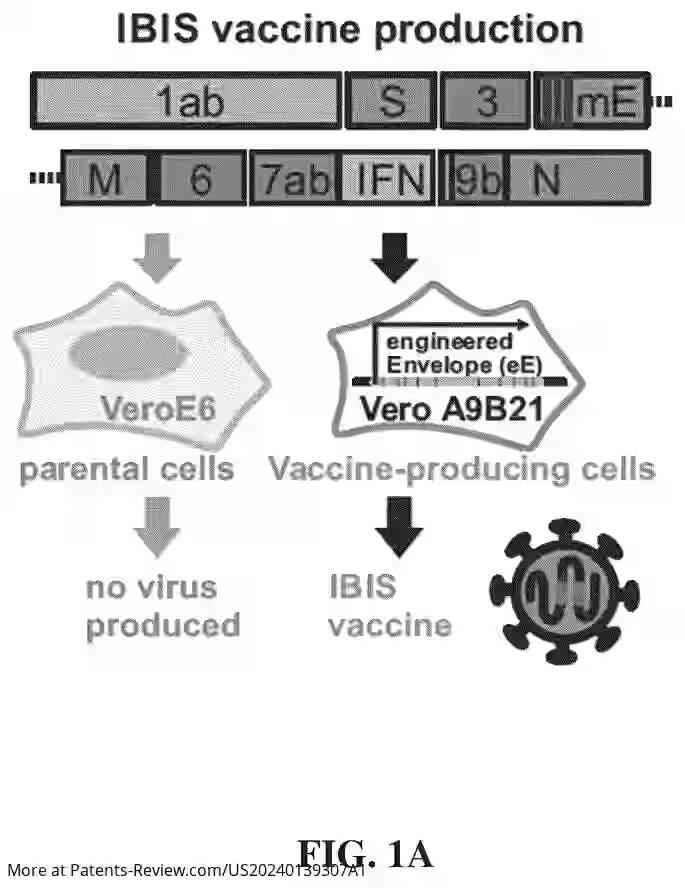
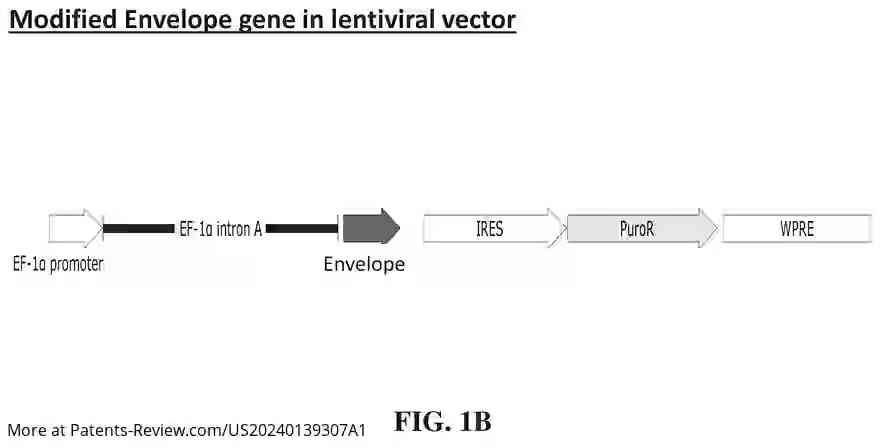
The vaccine construct involves a modified sarbecovirus genome featuring a modified envelope gene and an integrated nucleic acid encoding an interferon. This integration replaces viral open reading frames with type I interferons, such as IFNβ, enhancing immune responses. The modification includes stop codons in the envelope gene to inactivate the virus for safe use in vaccines. The approach targets a wide range of sarbecoviruses, including multiple SARS-CoV-2 variants and related viruses.
Applications and Benefits
The vaccine is designed for different administration routes, including mucosal (nasal spray) and parenteral methods (intradermal or intramuscular). It serves multiple purposes: vaccination, prophylactic immunization, prevention of infection contraction and transmission, reduction of infection severity, and treatment of existing infections. The vaccine's ability to enhance both B and T cell immunity positions it as a promising next-generation solution against evolving sarbecoviruses.
Research Findings
Research demonstrated that this universal sarbecovirus construct significantly reduces infection and transmission of SARS-CoV-1 and SARS-CoV-2. When administered post-infection, it lessens severity across variants like alpha, delta, and omicron. The incorporation of IFNβ notably boosts mucosal immune responses, which is crucial for combating respiratory viruses. This broad-spectrum immunity is achieved through enhanced protective B and T cell responses.
Conclusion
This patent application presents a groundbreaking approach to vaccine development by integrating interferon into the viral genome. It addresses the rapid mutation challenges posed by sarbecoviruses by offering a universal solution that adapts to various strains. The invention promises significant advancements in preventing and treating infections caused by these viruses, with potential applications in creating comprehensive pharmaceutical compositions and treatment kits.