SOLID DOSAGE FORM PRODUCTION
US20240148657
2024-05-09
Human necessities
A61K9/2095
Inventor:
Assignee:
Applicant:
Drawings (4 of 36)


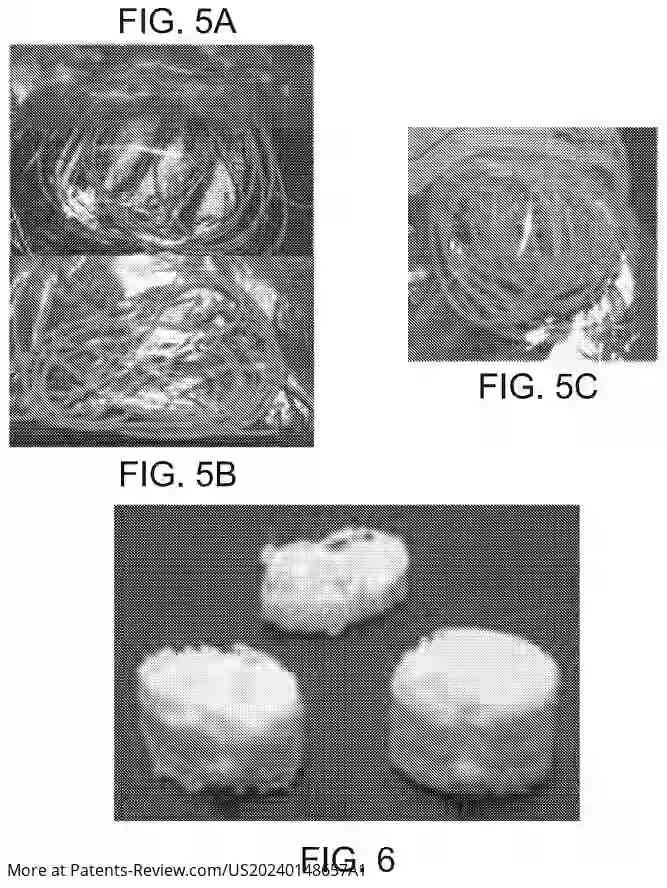
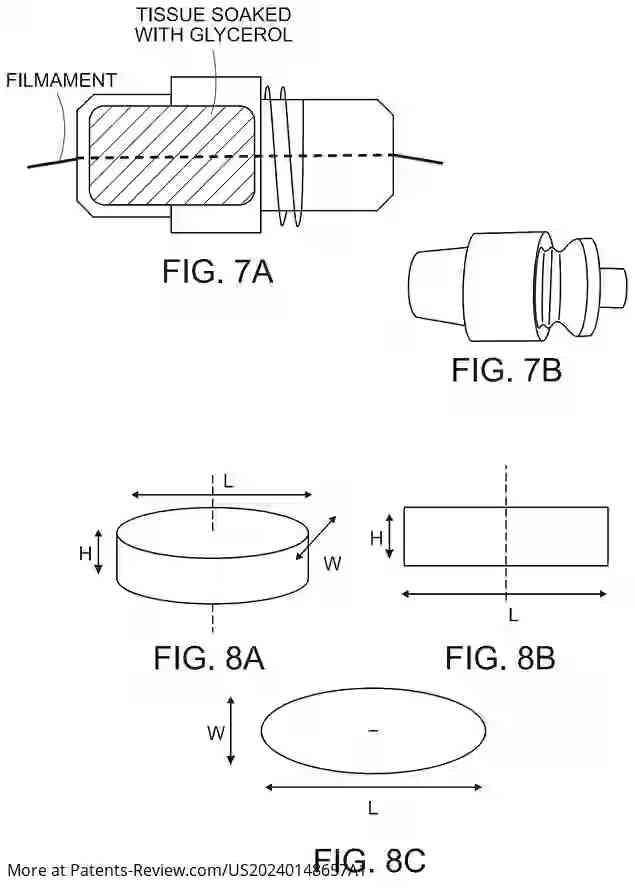
Smart overview of the Invention
The invention introduces a method for producing solid dosage forms, such as pharmaceutical tablets, using fused filament fabrication (FFF) 3D printing technology. This process leverages innovative printing filaments containing active ingredients, enabling the creation of personalized medicines directly from a 3D printer. The approach facilitates in situ production at locations like pharmacies, offering tailored treatments for patients. Additionally, the invention encompasses specialized software for printer operation and systems for monitoring multiple printers globally to ensure regulatory compliance and detect malfunctions efficiently.
Components and Methodology
The solid dosage form printing apparatus comprises several key components:
- A fused filament fabrication (FFF) 3D printer.
- A build platform for constructing the solid dosage form.
- Active ingredient-containing printing filaments, which may include carriers or precursors transforming into active ingredients during printing.
- Optional additional filaments with varying compositions.
- A computer for controlling the 3D printer and build platform, potentially connected to databases and specialized software.
The method involves providing this apparatus, operating the printer to create the dosage form on the platform, and optionally performing further processing steps like coating or modifying the form's properties.
Background and Challenges
Solid dosage forms are preferred due to their ease of administration and stability. However, traditional manufacturing methods are complex and costly, limiting customization. Previous attempts at using 3D printing for drug production faced challenges like regulatory approval difficulties, limited drug solubility in ink solutions, and high complexity due to numerous ingredients. This invention aims to overcome these obstacles by simplifying the process with fewer variables and improving stability through solid filaments.
Objectives
The invention seeks to address several issues inherent in prior art by providing:
- Methods for producing customizable solid dosage forms on-demand with minimal complexity.
- Enhanced storage stability for input materials and products.
- A system suitable for regulatory approval usable in pharmacies or approved facilities.
- The ability to aesthetically customize forms without compromising integrity.
- A tool for prototyping and formulation research in the pharmaceutical industry.
Applications and Benefits
This approach allows for high-dose active ingredient formulations with customizable drug release profiles. It minimizes input variables, reducing errors and enhancing quality control. By enabling real-time monitoring and data collection, it supports regulatory compliance and facilitates easier approval processes. The technology holds potential for widespread use in personalized medicine production, offering significant advancements over traditional manufacturing techniques.