GENE EDITING WITH A MODIFIED ENDONUCLEASE
US20240150796
2024-05-09
Chemistry; metallurgy
C12N15/907
Inventors:
Applicant:
Drawings (4 of 11)
Smart overview of the Invention
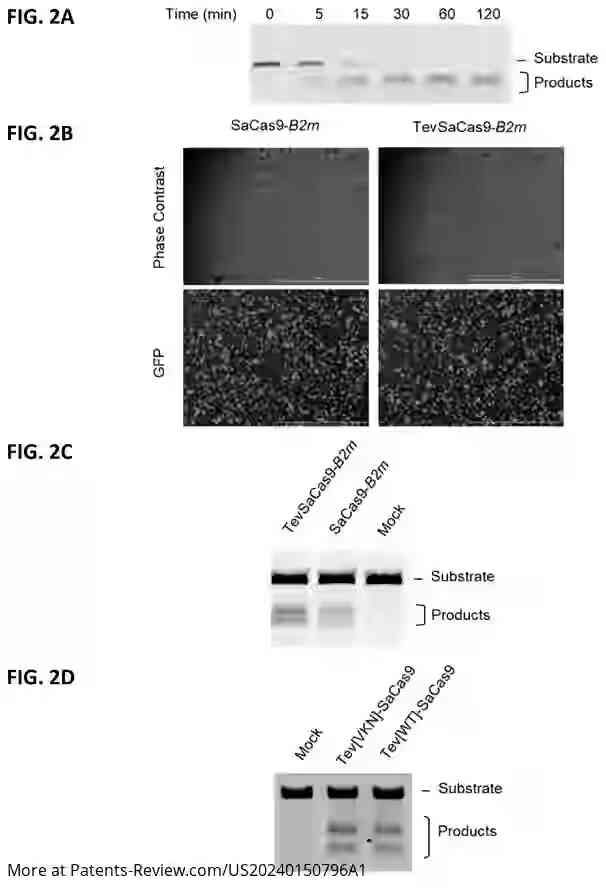
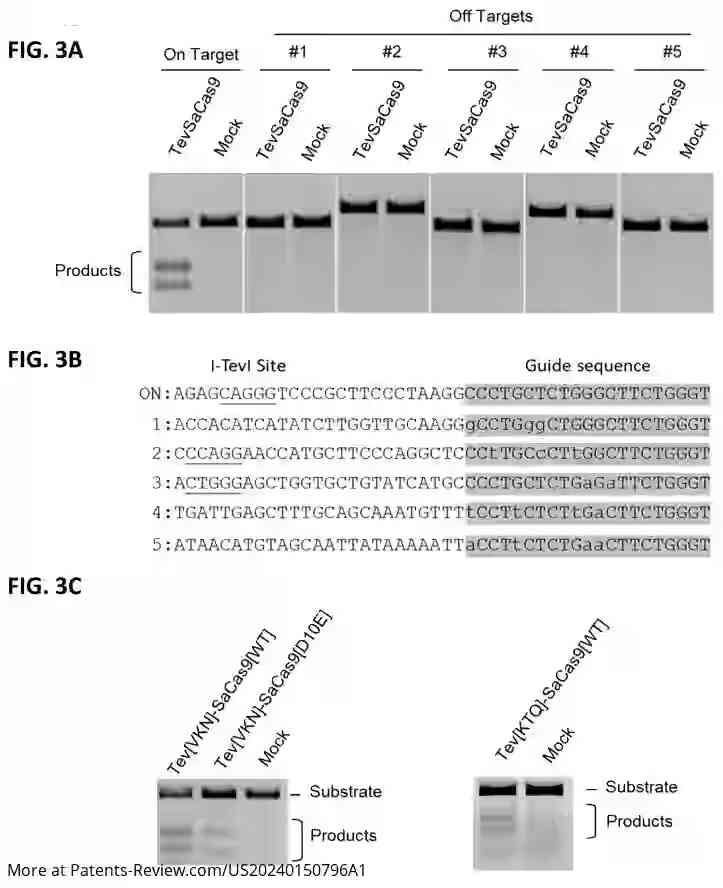
The patent application discusses a novel gene-editing technology utilizing a chimeric nuclease known as TevSaCas9. This engineered nuclease combines elements from the I-TevI domain and the Staphylococcus aureus Cas9 domain, aiming to enhance the precision and efficacy of gene editing in cell therapies. The technology is designed to address limitations of existing gene-editing tools by improving specificity and reducing off-target effects, crucial for therapeutic applications.
Background
Cell therapies are pivotal in treating various diseases, including cancer, by using healthy human cells to regenerate tissues or produce essential enzymes. Traditional treatments like chemotherapy have significant side effects, whereas immunotherapy leverages the body's immune system for targeted action. Gene editing plays a central role in these therapies by modifying immune cells to attack cancer cells or repairing genetic defects. Current technologies, such as CRISPR-Cas9, face challenges related to specificity and safety, which this invention seeks to overcome.
Technical Details
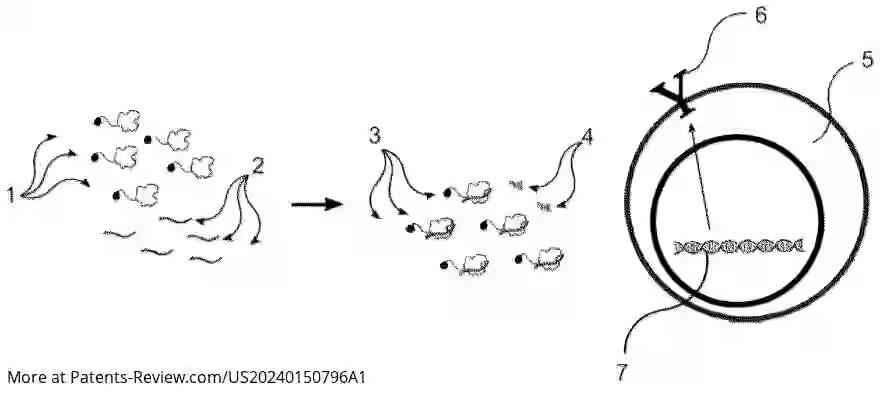
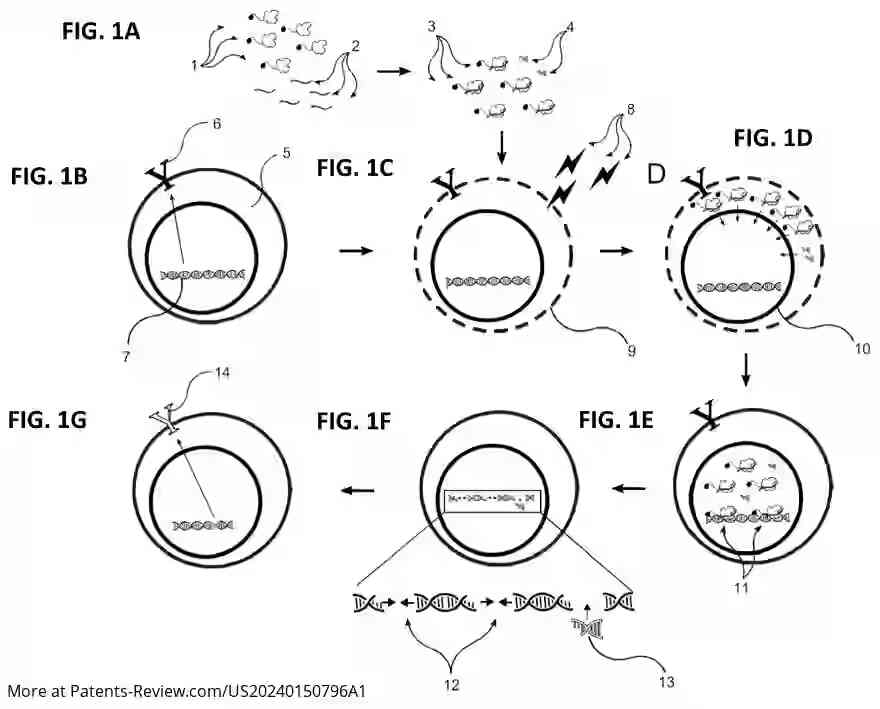
The TevSaCas9 nuclease is engineered to target specific genes involved in immune response and disease processes, such as B2M, TRAC1, TRCB1, and HLA-A. It can perform precise DNA modifications either by replacing sequences with donor DNA or deleting sequences without it. The invention also includes methods for creating genetically engineered cells using this nuclease, which can be applied ex vivo to immune cells or pluripotent stem cells.
Applications
This technology is applicable in creating advanced cell therapies by modifying chimeric antigen receptors (CARs) on T cells. These modifications can enhance the effectiveness of CAR-T cell therapies used in cancer treatment by reducing cell exhaustion and toxicity. The invention also allows for multiplexed gene disruption, targeting multiple genes simultaneously to improve therapeutic outcomes.
Implementation
The application outlines methods for delivering the TevSaCas9 nuclease into target cells without using viral vectors, thus improving safety profiles. Electroporation is suggested as a delivery method, allowing efficient integration of donor DNA at specific genomic sites. This approach aims to provide a more controlled and precise gene-editing process compared to traditional methods.