METHOD FOR INDUCING CELLULAR IMMUNITY TO INFECTION BY SARS-CoV-2
US20240165220
2024-05-23
Human necessities
A61K39/215
Inventor:
Assignee:
Applicant:
Drawings (4 of 13)
Smart overview of the Invention
The patent application introduces a plasmid DNA (pDNA)-based vaccine designed to induce cellular immunity against SARS-CoV-2, the virus responsible for COVID-19. This innovative vaccine targets the spike (S) protein of SARS-CoV-2, aiming to provoke both humoral and cellular immune responses. Unlike existing mRNA vaccines that require encapsulation and cold storage, this pDNA vaccine is noted for its thermostability and ease of production, potentially offering a more practical and cost-effective solution for widespread vaccination efforts.
Background
SARS-CoV-2, like other coronaviruses such as SARS-CoV and MERS-CoV, utilizes its spike protein to enter host cells by binding to the ACE2 receptor. The spike protein is crucial in eliciting an immune response, making it a primary target for vaccine development. Current vaccines, including mRNA and adenovirus vector types, have limitations such as storage requirements and potential side effects. The pDNA vaccine approach seeks to overcome these challenges by offering a stable and scalable alternative.
Advantages of pDNA Vaccines
Plasmid DNA vaccines present several benefits over traditional methods. They are safer, more economical, and can be produced without the need for high-level biosafety facilities. Their stability at various temperatures simplifies logistics compared to mRNA vaccines that require cold chain management. Moreover, pDNA vaccines do not carry the risk of infection or reversion associated with live virus vaccines, as they use only a gene segment to stimulate immunity.
Technical Details
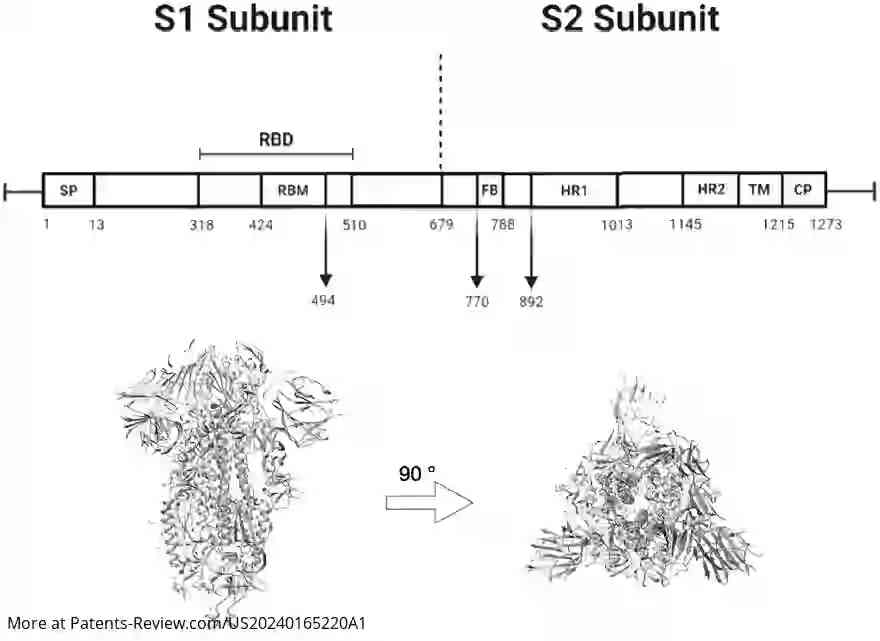
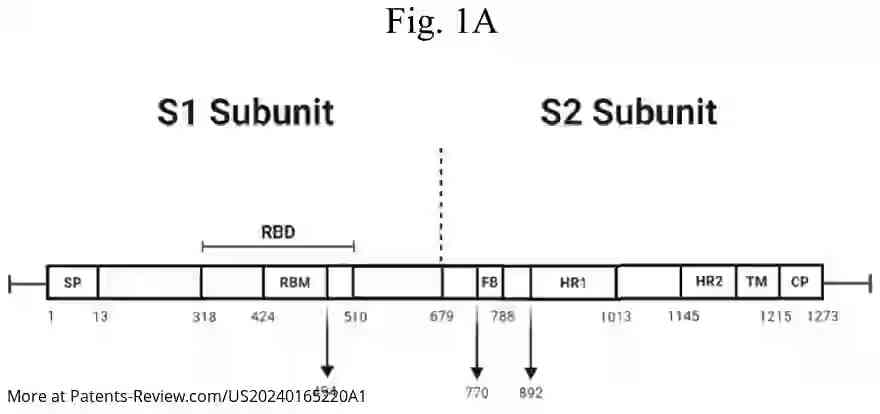
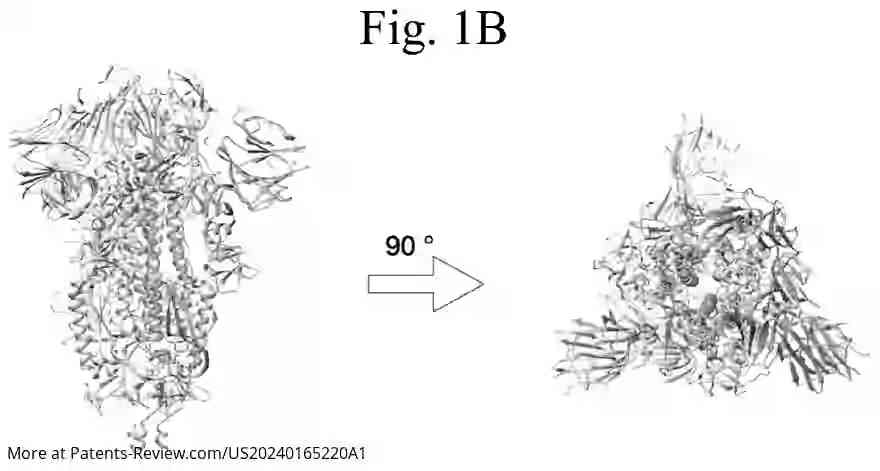
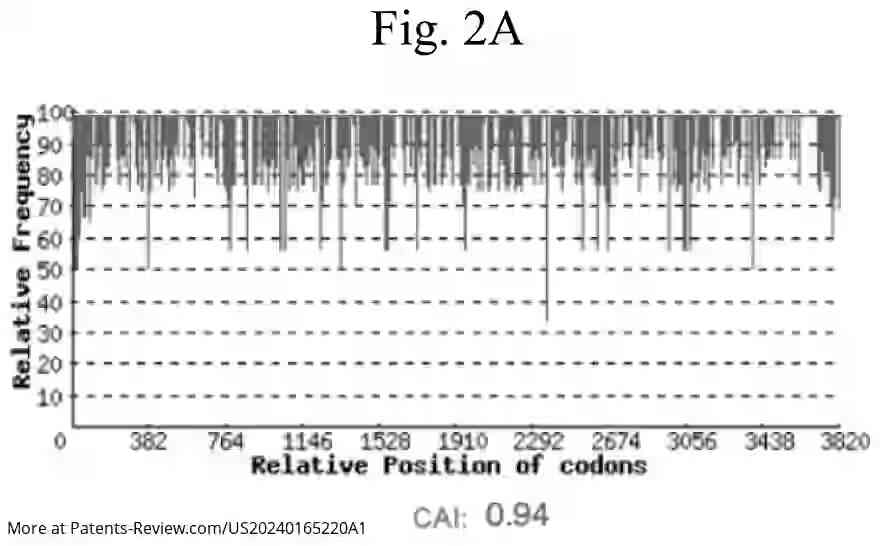
The pDNA vaccine specifically encodes segments of the SARS-CoV-2 spike protein, particularly the S1 subunit, which includes the receptor-binding domain essential for viral entry into cells. The application details various embodiments of the vaccine, including sequences that are highly identical to specific reference sequences (SEQ ID NO: 1 or 3), ensuring robust immunogenic response. Modifications in codon usage and CpG site distribution are employed to enhance vaccine efficacy.
Potential Implications
This pDNA-based approach could significantly impact global vaccination strategies by providing a more accessible and durable solution against COVID-19. Its scalable production process and stable storage conditions make it especially suitable for deployment in regions lacking advanced infrastructure. Furthermore, its ability to induce both humoral and cellular immunity positions it as a promising candidate in the ongoing fight against SARS-CoV-2 and potential future coronavirus outbreaks.